A new project to examine if the diet supplement developed in a Royal Holloway lab is suitable to treat epilepsy during pregnancy

New research led by Professor Robin Williams will investigate whether an innovative dietary supplement, developed from research completed in his lab, is suitable for use during pregnancy. The findings could provide the crucial evidence needed to offer women with epilepsy more treatment options to help them manage seizures during pregnancy.
“There is still far too little research into how epilepsy treatments affect women and their unborn babies during pregnancy – leaving many people without evidence-based treatment options,” says Professor Williams. “There is a clear and urgent need to develop treatments that have been thoroughly tested in pregnancy and shown to be safe.”
The new pregnancy study
Every year roughly 2,500 babies are born to mothers with epilepsy. Although most of these pregnancies proceed normally, the threat of epileptic seizures poses a serious risk – from reducing oxygen to the baby to increasing the chance of injury to the mother. On top of this, the long-term treatments currently used for epilepsy, such as anti-seizure medications and the ketogenic diet, can have potential side effects that could affect the development of the baby, leaving parents facing difficult choices.
“Based on what we know about how the new diet supplement works, it is expected to be safe for the developing baby, but more research is needed to provide the necessary evidence to confirm this.”
Researchers will examine the effects of the dietary supplement using laboratory models and brain cells grown in dishes. They’ll compare these results to the effects of a current high-risk anti-seizure medication and the effects of ketones, the molecules produced from fats in the ketogenic diet - which is currently the standard way to treat drug-resistant epilepsy.
The new funding from Action Medical Research will see Robin and his research team collaborate with researchers at UCL and the University of Edinburgh to carry out the laboratory studies.

Treating epilepsy
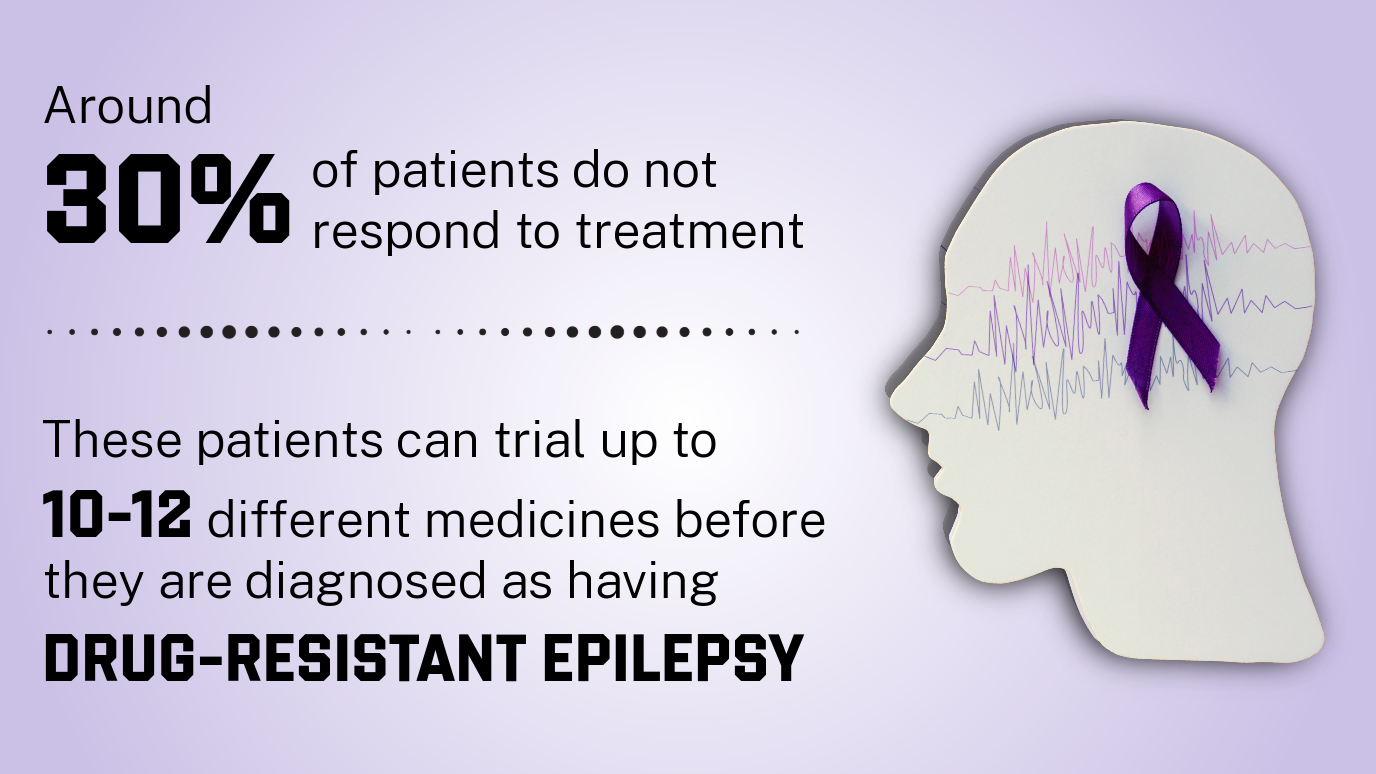
Epilepsy is the most common neurological disease after migraine, affecting around 1 in 100 people in the UK, and 50 million people worldwide. While existing medicines work well for many, around 30% of patients do not respond to the treatment and who, after trialing as many or 10 to 12 different drugs, are eventually diagnosed as having the drug-resistant form of the disease.


For these patients the only opportunity to find relief from the debilitating symptoms is to try to control the seizures through diet. The main alternative to the drugs is the ketogenic diet – a strictly controlled diet with a limited variety of food that is difficult for many people to maintain. “Imagine being told your entire day’s carbohydrate allowance is half a piece of bread every day for the rest of your life,” Robin says. “Adults don’t like it, and most just can’t follow it.”
This treatment option also involves additional NHS resources and a supporting team of specialists, including regular follow up appointments and monitoring by a nutritionist, creating another reason why clinicians may be reluctant to prescribe it.
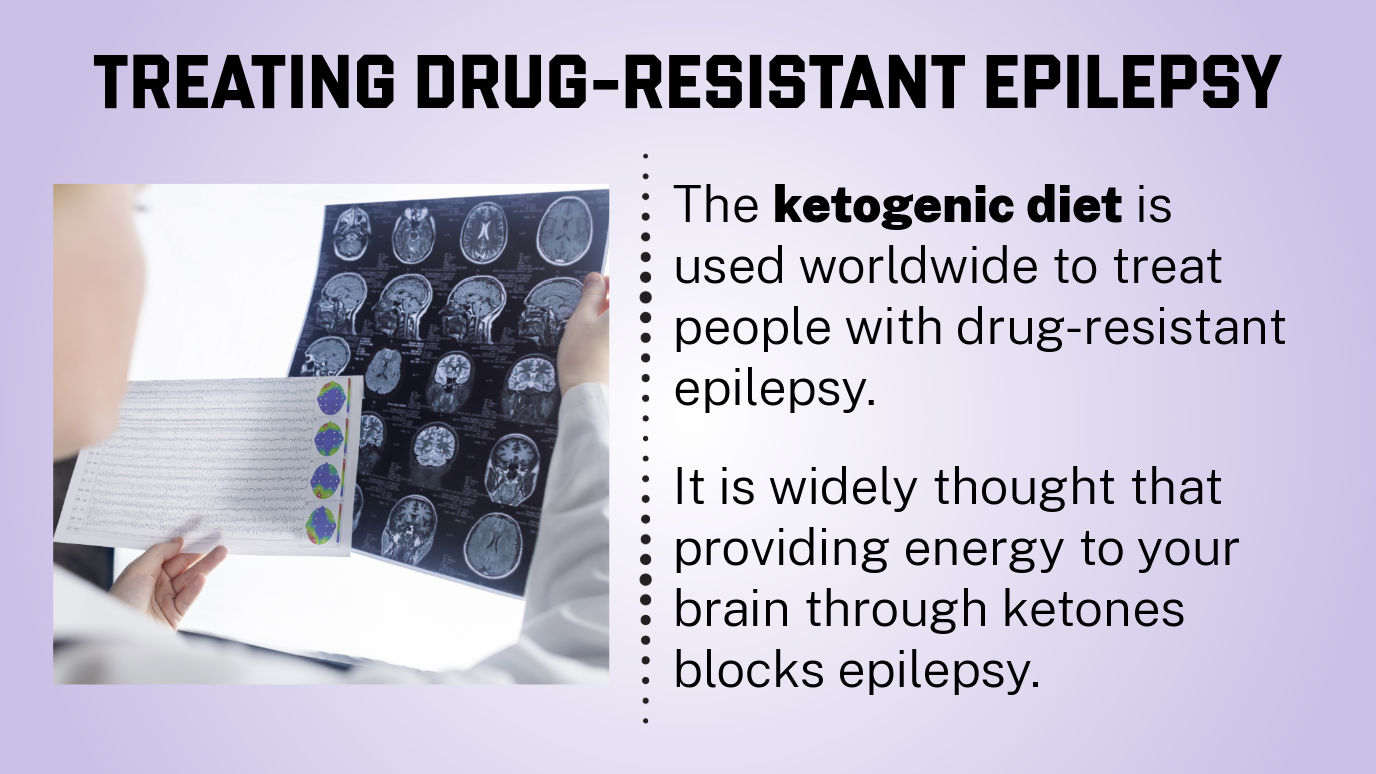
What is a ketogenic diet? And how was the dietary supplement developed
The ketogenic diet is used worldwide to treat people with drug-resistant epilepsy.
What is a ketogenic diet? |
|
A diet very low in carbohydrates and very high in fat, • • • • • • • • • |
Until now, it was widely thought that providing energy to your brain through ketones helps control epileptic seizures. But Robin and his team discovered that it may not be how the ketogenic diet works for people with epilepsy.
While studying the common epilepsy drug sodium valproate, he and his team identified several chemicals that regulate fatty acids in cells, including decanoic acid. This caught their attention. Decanoic acid is a key part of the MCT (medium chain triglyceride) diet, a version of the ketogenic diet prescribed for those with drug-resistant epilepsy. Most people believed that it worked after being turned into ketones, but the new research indicated that it was the direct effects of decanoic acid itself.
"We started thinking, maybe the MCT diet doesn't work by ketones - it works by decanoic acid having its molecular effect."
Working with clinicians and industry partners, they developed a new decanoic acid-rich dietary supplement and tested it with a small group of people whose epilepsy could not be controlled with medication. “It worked really well” says Robin. “And, because the treatment doesn’t rely on producing ketones, patients can follow a normal diet and simply take the supplement alongside it.”
Building on the success of the trials, this new treatment has now been developed into K.Vita, a commercial product already in clinical use across the UK and in parts of Europe.

Why this research matters
For Robin, the motivation is simple: Science should improve lives.
“I think science should be there to improve our society, our well-being, and our health - to help us live happier and healthier lives.”
Although some might say his lab is focused on ‘blue-sky’ research, his drive to find real-world applications for his discoveries shines through. The impact of his research can be seen in the partnerships and collaborations he establishes, and the new treatments developed that are changing patients’ lives.
“Through using an amoeba, we have developed a dietary treatment that is now in use by the NHS to treat babies with drug resistant epilepsy. I'm really proud of that and I think scientists should continue to focus on real-world impact to improve quality of life.”
As the new pregnancy study begins, Robin and his team are also exploring how the dietary treatment could help other hard to treat and rarer forms of epilepsy. Early research, using patient-derived stem cells from a rarer drug-resistant epilepsy linked with movement disorders, is showing promising results and offering hope to families and patients dealing with the most challenging epilepsies.

Robin in his lab



Learn more about the International Neurological Ketogenic Society via their website:
INKS homepage
Return to our Research in Focus page to uncover more exciting research happening at Royal Holloway, University of London.
Research in Focus homepage